This worksheet is intended for upper middle s. When atoms are formed from protons, neutrons, and electron, some mass is converted into energy, called the binding energy. In this Atomic Structure Worksheet, students are asked to recall all of the information found in an element square, sketch Bohr Diagrams of atoms, calculate the number of neutrons and valence electrons in an atom, and create Lewis/electron-dot diagrams. We cannot use these data to calculate the mass of a given atom, because the mass of a nuclide is not simply the sum of the masses of its fundamental particles. In atomic mass units the fundamental particles have the following masses Thus, atom consists of a heavy positively charged nucleus in the centre containing all protons and the electrons were revolving around the nucleus so that the centrifugal force balances the force of attraction.Ītomic number and mass number: The general notation that is used to represent the mass number and atomic number of a given atoms is $_\] It was therefore, concluded that there was sufficient empty space within the atom and small heavy positively charged body at the center called nucleus. 
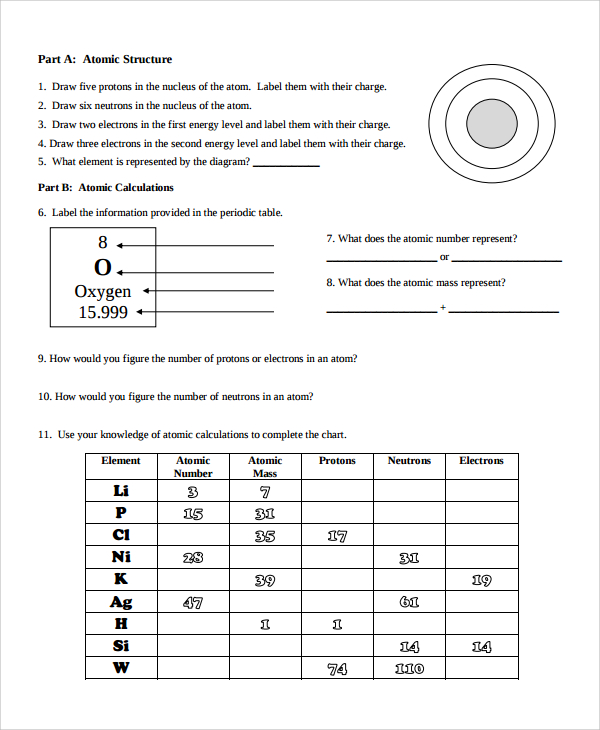
Most of the particles passed through the foil undeflected, a few were deflected through small angle while very few were deflected back. Draw 2 electrons in the 1st energy level and label with their charge. Draw 6 neutrons in the nucleus and label with the charge. Rutherford’s model of atom: A thin foil of gold was bombarded with -particles. Atomic Structure: Name: Date: B: Sketch An Atom Draw 5 protons in the nucleus and label with the charge. Rutherford’s -rays scattering experiments. This Atomic Structure practice worksheet is perfect for students to exhibit mastery on atomic structure for both neutral atoms and isotopes. us Chapter 4 Atomic Structure Guided Practice Problems Answers Chapter 4. Web certain topics in this worksheet should be review, such as the idea of protons, neutrons, and electrons, while other topics, such as atomic mass units and the electron cloud, are. Click the card to flip Protons Click the card to flip 1 / 15 Flashcards Learn Test Match Created by shadow7 Terms in this set (15) 1. Thomson’s model of atom: An atom is a sphere of positive electricity in which sufficient number of electrons were embedded to neutralize the positive charge just as seeds in a melon or raisins in pudding. Chapter 4 Worksheet 1 - Key An atom has 19 protons 19 electrons and 20 neutrons. Web atomic structure worksheet answers key. Atomic Structure Worksheet 5.0 (1 review) Get a hint 1. Neutron: A fundamental particle which carries no charge but has a mass nearly equal to that of hydrogen atom. CHEMISTRY ATOMIC STRUCTURE PRACTICE I 17 Isotopic Symbol NaPart B. Proton: A fundamental particle which carries one unit positive charge and has a mass nearly equal to that of hydrogen atom. IsotopeAtomic Structure Practice worksheet Live worksheets > English > Science > Atoms. 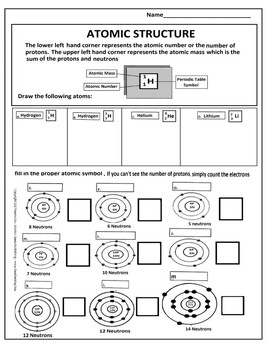 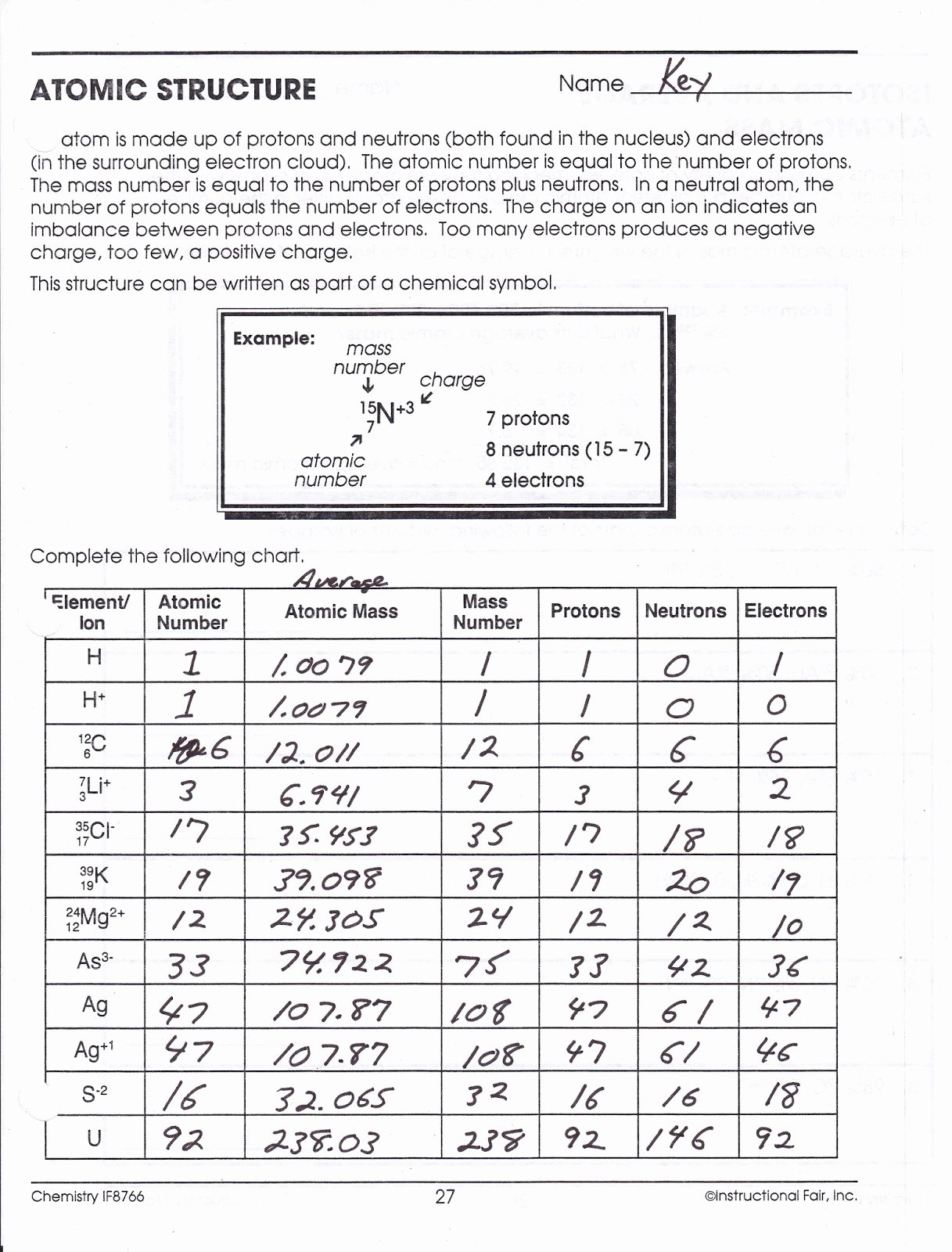
It is made up of electrons, protons and neutrons called fundamental particles.Įlectron: A fundamental particle which carries one unit negative charge and has a mass nearly equal to 1/1837th of that of hydrogen atom. Structure of Atom Chapter at a Glance - Class 11 NCERT SolutionsĬonstituents of atom: Atom is no longer considered as indivisible. You can also download the Structure of Atom Class 11 questions and answers PDF, and go through them. Our subject matter experts will get the right answers for all the questions that are asked in the examinations relating to Class 11 Chemistry Chapter 2. The Class 11 Chemistry NCERT Solutions Chapter 2 are prepared by the experts of Vedantu who are working in this field for decades now. NCERT Solutions for Class 11 Chemistry Chapter 2 are available for you to download online in PDF format. Drawing of atomic model: What he discovered: Equipment used.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
